Industries
Health Care
Plastic injection molding for consumer health care products — wearables, home diagnostics, personal care devices, and wellness monitors. Biocompatible materials, clean room available.
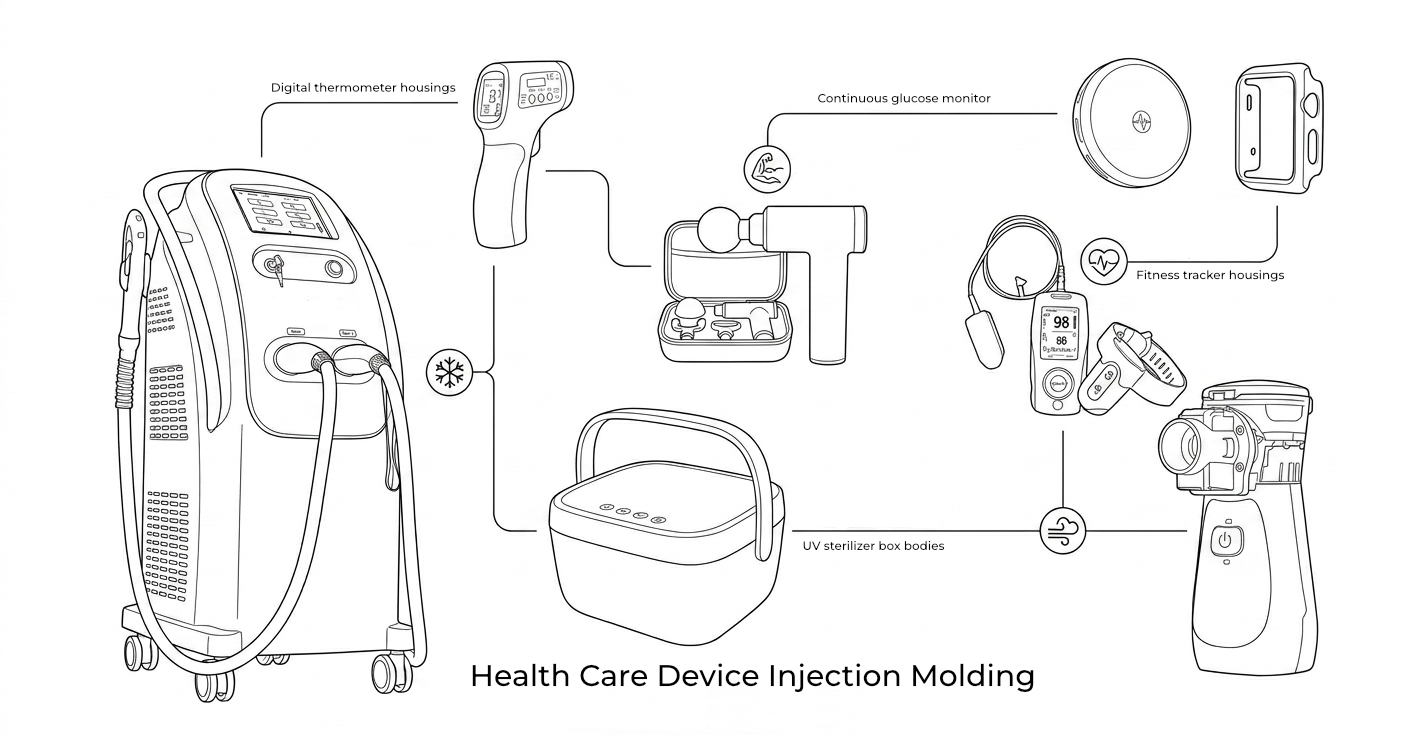
Health Care Device Injection Molding
Consumer health care products bridge the gap between medical devices and consumer electronics — they are worn on or near the body, must pass regulatory scrutiny, and need the cosmetic quality consumers expect from premium lifestyle products.
JBRplas manufactures plastic enclosures and components for wearable health devices, home diagnostics, personal care appliances, and wellness monitors. Our experience spans skin-contact rated materials, soft-touch overmolding for comfortable wear, and IP-rated enclosure designs for devices used in humid environments.
Health Care Segments We Serve
Wearables & Monitoring
- Smartwatch cases and wristband components
- Fitness tracker housings and clip bodies
- Continuous glucose monitor (CGM) patch holders
- Heart rate and SpO₂ monitor enclosures
- Sleep monitoring device housings
- ECG patch and electrode holder components
- Blood pressure cuff bodies and display housings
- Posture correction device shells
Home Diagnostics
- Digital thermometer housings (oral, ear, forehead)
- Blood glucose meter cases and lancing device bodies
- Pulse oximeter enclosures
- Pregnancy and ovulation test device holders
- Hearing screening device housings
- Vision test peripheral components
- Allergy testing kit housings
Personal Care Appliances
- Electric toothbrush bodies, necks, and head housings
- Oral irrigator bodies and reservoir components
- Electric shaver and grooming device housings
- Hair removal device bodies (IPL, laser)
- Skin analysis device enclosures
- UV sterilizer box bodies and lids
Wellness & Rehabilitation
- TENS / EMS therapy unit housings
- Heat therapy pad covers and controllers
- Massage device shells and heads
- Portable nebulizer and inhaler housings
- Light therapy panel frames
- Hearing aid outer shells and battery door components
Critical Material Requirements
Skin-Contact Biocompatibility
Devices worn against skin require materials that do not cause irritation, sensitization, or cytotoxicity. JBRplas works with the following skin-contact rated resins:
| Material | Certification | Key Properties |
|---|---|---|
| ABS (medical grade) | USP Class VI | Rigid enclosures, housings |
| PC (medical grade) | USP Class VI / ISO 10993 | Optical clarity, impact resistance |
| PP (medical grade) | USP Class VI / FDA | Chemical resistance, lightweight |
| TPE / TPU (medical) | ISO 10993 | Soft-touch, bands, gaskets, overmolds |
| PEEK | ISO 10993, USP Class VI | High-performance, sterilizable |
All biocompatible resins used at JBRplas are supplied with manufacturer compliance certificates. Lot numbers are traceable per production batch to support regulatory submissions.
Overmolding for Wearable Comfort
Many wearable health devices require a rigid structural substrate overmolded with a soft, skin-friendly material:
- Wristbands and straps — PC or ABS substrate overmolded with medical-grade TPE
- Body-contact pads — soft TPU overmolded onto structural clips or holders
- Grip surfaces — TPE overmolding on handheld diagnostic device bodies
- Sealing gaskets — integrated TPE seal overmolded into housing for IP rating
JBRplas designs and manufactures both the substrate mold and the overmold tool, ensuring material compatibility and interface geometry are engineered correctly from the start.
IP Rating Enclosure Design
Devices used in bathrooms, during exercise, or in clinical environments typically require IPX4 (splash) to IPX7 (1m submersion) ratings. IP-rated enclosure design requires:
- Precision-fit parting surfaces with integrated TPE gasket channels
- Ultrasonic weld joint design for hermetic assembly
- Careful management of button and port apertures
- Tolerance control at all mating interfaces: ±0.05mm or better
Our DFM review specifically addresses IP rating achievability based on your enclosure geometry and target IP class.
Regulatory Considerations
Consumer health care products vary widely in regulatory classification:
General wellness devices (fitness trackers, thermometers, sleep monitors) — typically require no 510(k) or CE MDR clearance but must meet applicable safety standards (UL, CE, RoHS).
Over-the-counter medical devices (blood glucose meters, pulse oximeters, blood pressure monitors) — FDA Class II or equivalent; requires appropriate quality documentation from supply chain.
Prescription devices — stricter supply chain requirements; JBRplas can provide FAI reports, material certificates, and PPAP-style documentation packages on request.
Regardless of device classification, JBRplas provides Certificate of Conformance, material certificates with lot traceability, and First Article Inspection reports as standard for all health care programs.
Other Industries We Serve
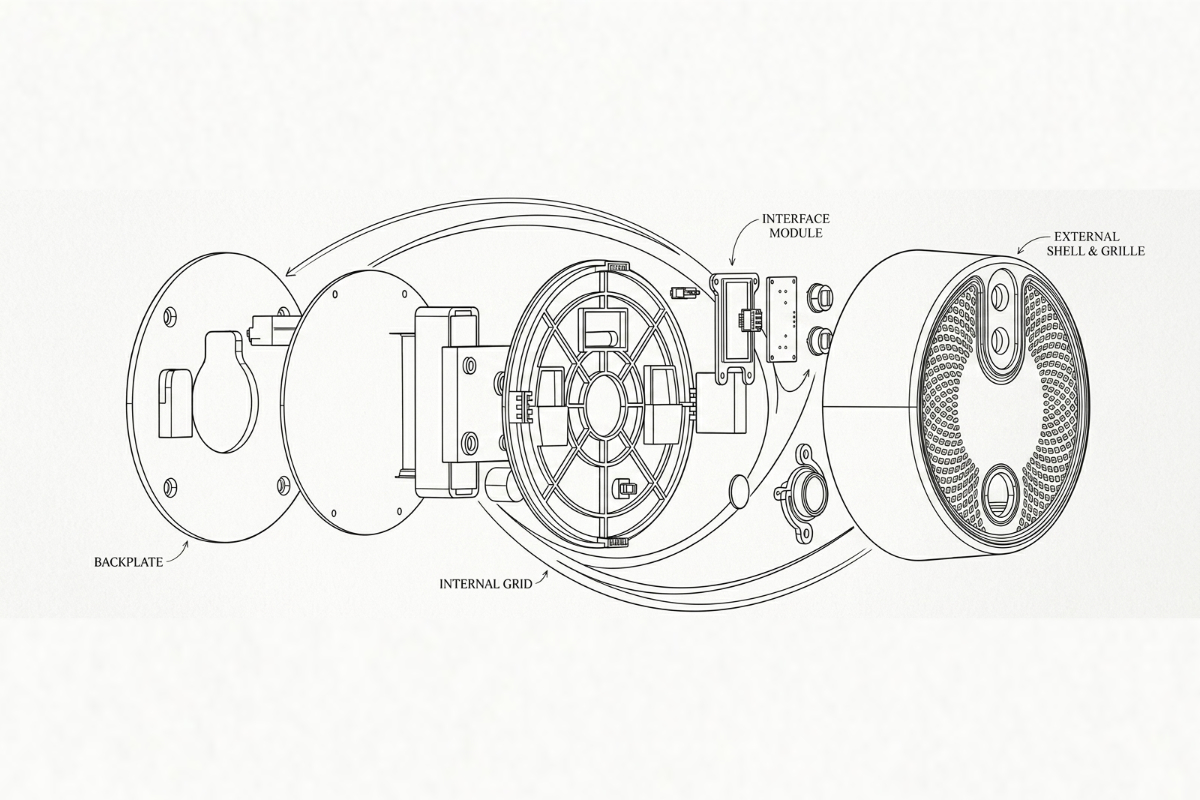
Plastic injection molding for smart home devices — hubs, sensors, switches, plugs, and cameras. UL94 V-0, Class A finish, high-volume production from Shenzhen.
Explore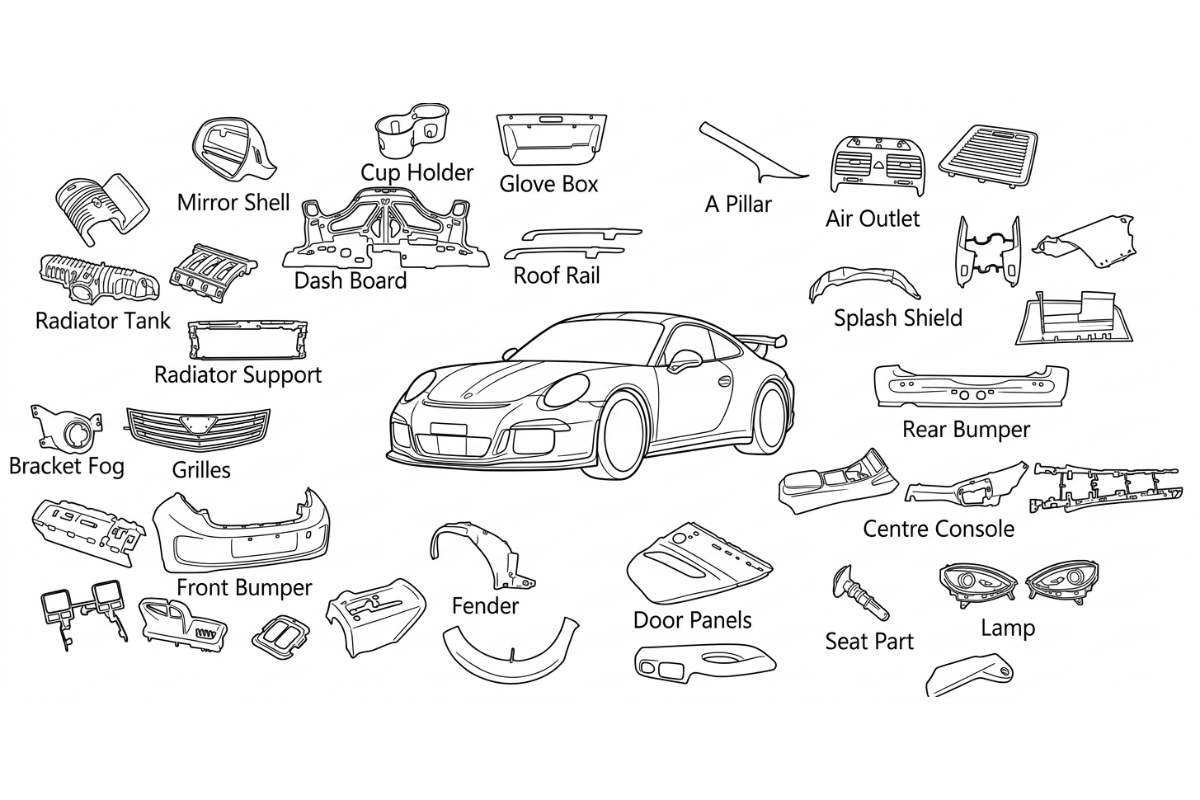
JBRplas manufactures precision plastic injection molds and molded parts for automotive OEM suppliers — interior trim, connector housings, HVAC components, and structural brackets.
Explore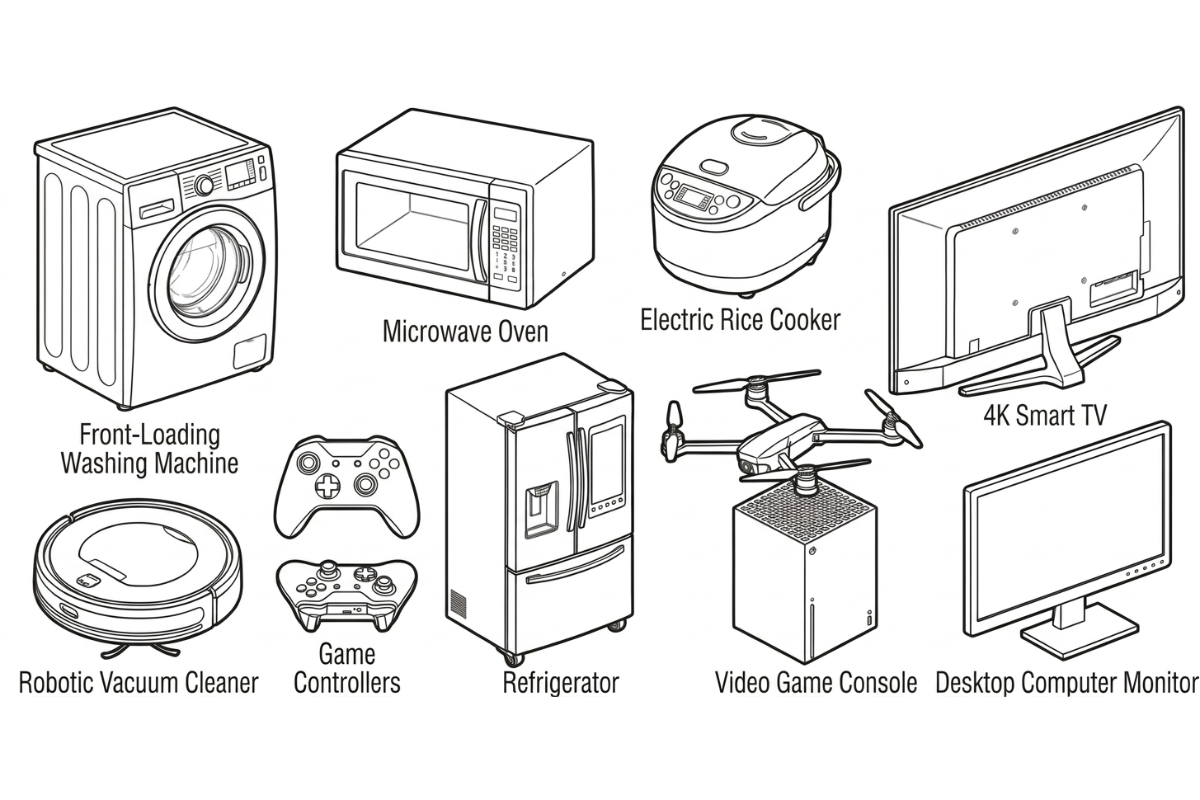
Plastic injection molded enclosures, housings, and components for consumer electronics and home appliances. Flame-retardant resins, UL94 V-0, Class A finish. Shenzhen factory.
Explore
JBRplas manufactures medical-grade plastic injection molds and molded components — FDA compliant resins, ISO 10993 biocompatible materials, clean room molding available.
Explore
Plastic injection molding for financial devices — POS terminals, card readers, ATM components, and payment kiosks. Precision tolerances, EMV compliance, tamper-evident designs.
Explore
Plastic injection molding for branded toys and children's products — EN71, ASTM F963, CPSC compliant. Vibrant colours, safe materials, high-volume production from Shenzhen.
Explore